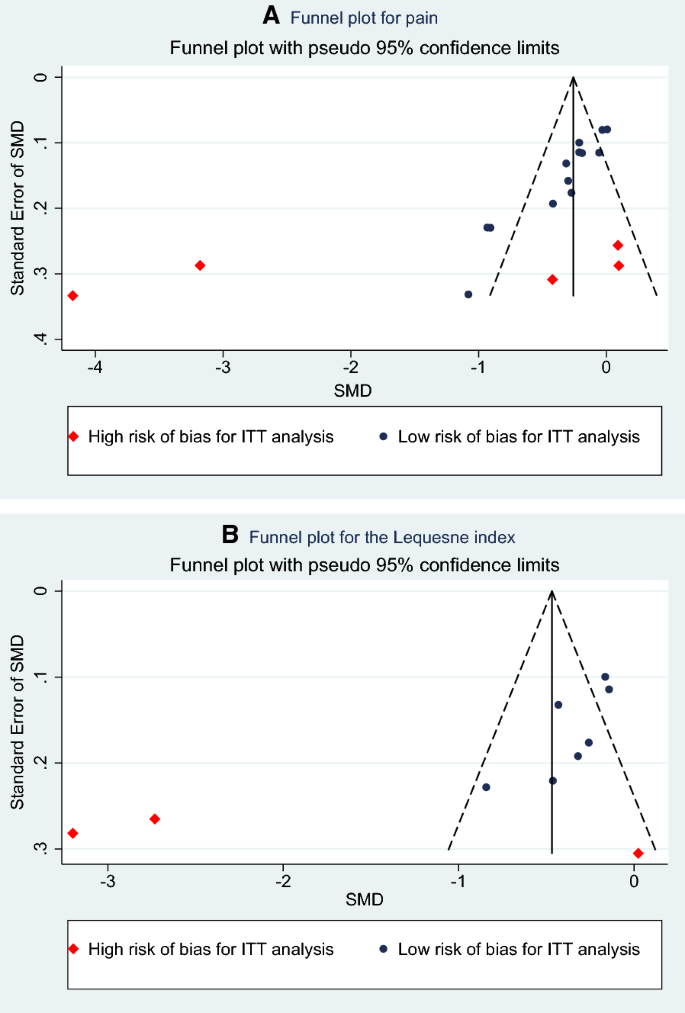
The need to arrive at decisions affecting clinical practise fostered the momentum toward "evidence-based medicine" 1 – 2. In many instances, the results of these multiple small studies of an issue are diverse and conflicting, which makes the clinical decision-making difficult. Important medical questions are typically studied more than once, often by different research teams in different locations. The need to integrate findings from many studies ensures that meta-analytic research is desirable and the large body of research now generated makes the conduct of this research feasible. Rigorously conducted meta-analyses are useful tools in evidence-based medicine. A failure to identify the majority of existing studies can lead to erroneous conclusions however, there are methods of examining data to identify the potential for studies to be missing for example, by the use of funnel plots. The specification of the outcome and hypotheses that are tested is critical to the conduct of meta-analyses, as is a sensitive literature search. The benefits of meta-analysis include a consolidated and quantitative review of a large, and often complex, sometimes apparently conflicting, body of literature. The examination of variability or heterogeneity in study results is also a critical outcome.

Outcomes from a meta-analysis may include a more precise estimate of the effect of treatment or risk factor for disease, or other outcomes, than any individual study contributing to the pooled analysis. Meta-analysis is a quantitative, formal, epidemiological study design used to systematically assess previous research studies to derive conclusions about that body of research. Methods used to produce a rigorous meta-analysis are highlighted and some aspects of presentation and interpretation of meta-analysis are discussed. The researchers also reported that Egger’s test showed no evidence that the results were significantly affected by publication bias.The objectives of this paper are to provide an introduction to meta-analysis and to discuss the rationale for this type of research and other general considerations. Improvements in sleep latency did not vary between individual hypnotics. Non-benzodiazepine hypnotics showed a small improvement in subjective sleep latency although the change was not significant (−6.9 minutes, −26.0 to 12.4). When the results of the trials were combined, non-benzodiazepine hypnotics showed a significant improvement (reduction) in the main outcome of polysomnographic sleep latency compared with placebo (weighted mean raw difference −22.0 minutes, 95% confidence interval −33.0 to −11.0). Thirteen trials were eligible for inclusion. The main outcomes were polysomnographic and subjective measurements of sleep latency. Participants were adults with primary insomnia (transient or chronic).
Randomised controlled trials were included if they were double blind, placebo controlled, and had parallel treatment groups.

Researchers undertook a meta-analysis of the effectiveness of non-benzodiazepine hypnotics, including eszopiclone, zaleplon, and zolpidem.

